 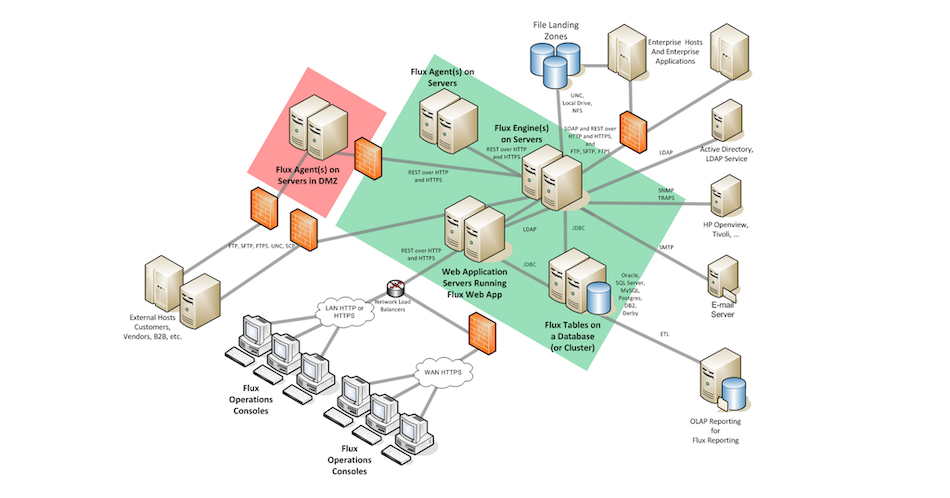
Most recent flux maps, such as those for developing embryos of oilseed rape ( Brassica napus Schwender et al., 2006 Junker et al., 2007) and sunflower ( Helianthus annuus Alonso et al., 2007a) or the one for a heterotrophic cell suspension of Arabidopsis ( Arabidopsis thaliana Williams et al., 2008), have well-defined mitochondrial fluxes. In principle, compartmented fluxes can be deduced from the labeling patterns of metabolites that are synthesized in a single compartment ( Ratcliffe and Shachar-Hill, 2005, 2006 Allen et al., 2007), and this property has been exploited with varying degrees of success in different studies. Subcellular compartmentation of steps and pathways can be incorporated into the model that describes the redistribution of the label, and flux maps of central carbon metabolism in plant cells typically aim to distinguish between fluxes in the cytosol, mitochondria, and plastids.  
In this approach, substrates labeled with stable isotopes are introduced into the network, and fluxes are determined by measuring the labeling of the system after it has reached an isotopic and metabolic steady state. The emergence of steady-state metabolic flux analysis (MFA) as a practicable systems biology tool for generating flux maps of the central metabolic pathways in plants offers new opportunities for analyzing plant metabolic phenotypes ( Ratcliffe and Shachar-Hill, 2006 Libourel and Shachar-Hill, 2008 Schwender, 2008 Kruger and Ratcliffe, 2009). The need to consider discrete pools of metabolites in specific compartments, and the transporters that link them, complicates the quest for a detailed, predictive understanding of the regulation of plant metabolism as a result, it remains difficult to manipulate flows of material through the central metabolic network in a predictable way ( Carrari et al., 2003 Kruger and Ratcliffe, 2008 Sweetlove et al., 2008). 
The Arabidopsis flux map also shows that the potential ATP yield of respiration in heterotrophic plant cells can greatly exceed the direct metabolic requirements for biosynthesis, highlighting the need for caution when predicting flux through metabolic networks using assumptions based on the energetics of resource utilization.Įxtensive subcellular compartmentation, with unique locations for many steps and pathways, as well as the duplication of other steps and pathways in different compartments, adds greatly to the structural complexity of the plant metabolic network ( Lunn, 2007 Kruger and Ratcliffe, 2008). The analysis emphasizes the inherent difficulty of analyzing the PPP without predefining the extent of its compartmentation and the importance of obtaining high-quality data that report directly on specific subcellular processes. The observed redistribution of the label could be explained by any one of three models of the subcellular compartmentation of the oxidative PPP, but other biochemical evidence favored the model in which the oxidative steps of the PPP were duplicated in the cytosol and plastids, with flux through these reactions occurring largely in the cytosol. Focusing on the pentose phosphate pathway (PPP), multiple data sets were fitted simultaneously to models in which the subcellular compartmentation of the PPP was altered. To address this question, fluxes through the pathways of central metabolism in a heterotrophic Arabidopsis ( Arabidopsis thaliana) cell suspension culture were deduced from the redistribution of label in steady-state 13C-labeling experiments using -, -, and glucose. Ideally, steady-state metabolic flux analysis, an emerging tool for creating flux maps of heterotrophic plant metabolism, would capture this feature of the metabolic phenotype, but the extent to which this can be achieved is uncertain. The presence of cytosolic and plastidic pathways of carbohydrate oxidation is a characteristic feature of plant cell metabolism. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
